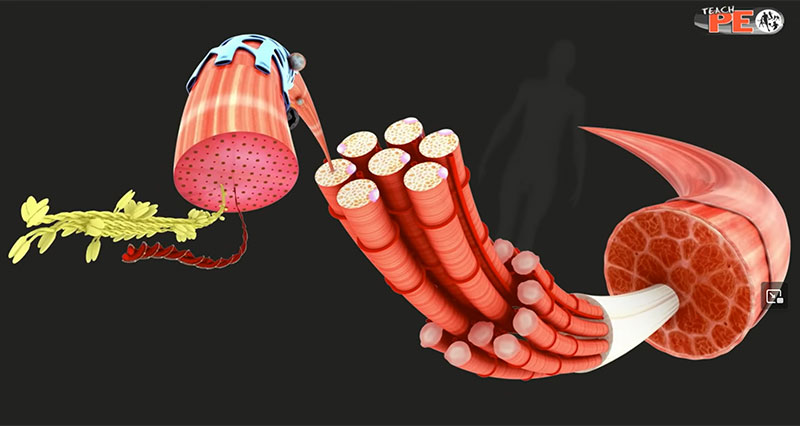
The sliding filament theory of muscle contraction is the mechanism by which muscles are thought to contract at a cellular level. It explains the steps in muscle contraction.
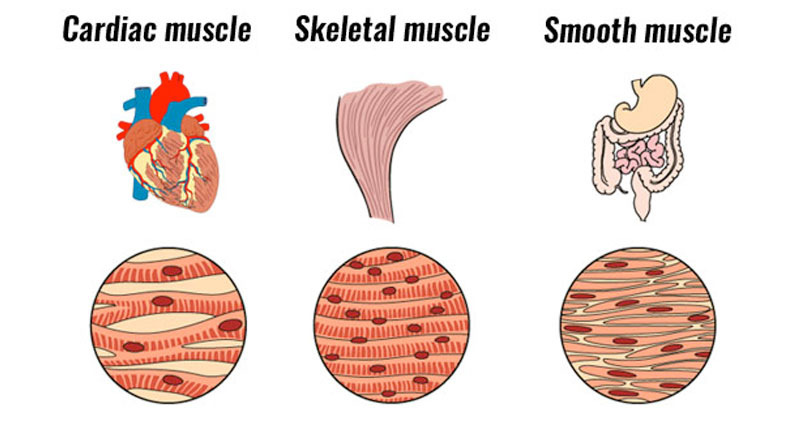
A good understanding of skeletal muscle structure is useful when learning how sliding filament theory works.
What is sliding filament theory?
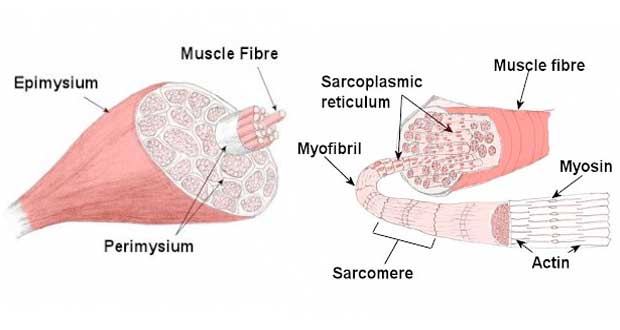
At a very basic level, each muscle fibre is made up of smaller fibres called myofibrils. These contain even smaller structures called actin and myosin filaments. These filaments slide in and out between each other to form a muscle contraction hence called the sliding filament theory!
The diagram above shows part of a myofibril called a sarcomere. This is the smallest unit of skeletal muscle that can contract. Sarcomeres repeat themselves over and over along the length of the myofibril.
Here is a quick reminder of the structure of muscle.
- Myofibril: A cylindrical organelle running the length of the muscle fibre, containing Actin and Myosin filaments.
- Sarcomere: The functional unit of the Myofibril, divided into I, A, and H bands.
- Actin: A thin, contractile protein filament, containing ‘active’ or ‘binding’ sites.
- Myosin: A thick, contractile protein filament, with protrusions known as Myosin Heads.
- Tropomyosin: An actin-binding protein that regulates muscle contraction.
- Troponin: A complex of three proteins, attached to Tropomyosin.
Steps in muscle contraction
Here is what happens in detail. We can explain muscle contraction in five parts.
1. Nerve impulse
A nerve impulse arrives at the neuromuscular junction, which causes a release of a chemical called Acetylcholine. The presence of Acetylcholine causes depolarisation of the motor endplate. Acetylcholine travels throughout the muscle by transverse tubules. As a result, Calcium (Ca+) releases from the sarcoplasmic reticulum.
2. Calcium binds to troponin
In the presence of high concentrations of Ca+, the Ca+ binds to Troponin, changing its shape and so moving Tropomyosin from the active site of the Actin. The Myosin filaments now attach to the Actin, forming a cross-bridge, enabling muscle contraction.
3. ATP breaks down
The breakdown of ATP releases energy which enables the Myosin to pull the Actin filaments inwards and so shortening the muscle. This occurs along the entire length of every myofibril in the muscle cell.
4. Myosin detaches
The Myosin detaches from the Actin and the cross-bridge is broken when an ATP molecule binds to the Myosin head. When the ATP is then broken down the Myosin head can again attach to an Actin binding site further along the Actin filament and repeat the ‘power stroke’. This repeated pulling of the Actin over the myosin is often known as the ratchet mechanism.
5. Muscle contraction
This process of muscular contraction can last for as long as there are adequate ATP and Ca+ stores. Once nerve impulses stop, Ca+ is pumped back to the Sarcoplasmic Reticulum. The Actin returns to its resting position causing the muscle to lengthen and relax.
It is important to realise that a single power stroke results in only a shortening of approximately 1% of the entire muscle. Therefore, to achieve a muscle contraction of up to 35% the whole process repeats many times. Whilst half of the cross-bridges are active pulling the Actin over the Myosin, the other half are looking for their next binding site.
Sliding filament theory diagrams
The following diagrams illustrate a stretched, partial, and full muscle contraction.
Stretched muscle contraction

The diagram above shows a muscle stretched. The I – bands and the H – zone is wider. The myosin and actin filaments overlap, but not by much. As a result, fewer cross bridges between Actin and Myosin form, so muscle strength is lower.
Partial muscle contraction

With a partial muscle contraction myosin and actin overlap more. Therefore, more potential for cross bridges to form. The I – bands and H – zone shortens.
Fully contracted muscle
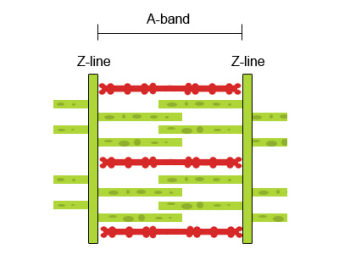
The diagram above shows a fully contracted muscle with lots of overlap between the actin and myosin. Because the thin actin filaments have overlapped there is a reduced potential for cross bridges to form again. Therefore, there will be low force production from the muscle.
External links